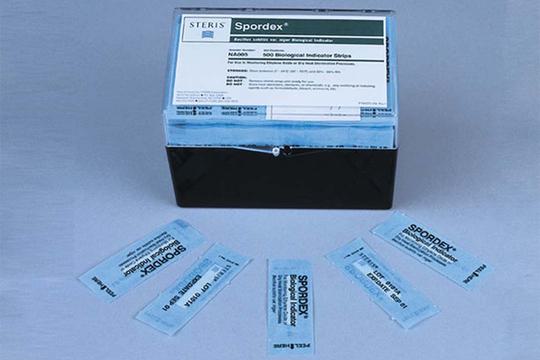
Autoclaves are used in various settings including hospitals, research labs, and manufacturing facilities to sterilize equipment, instruments, and other items. It is essential to validate autoclaves regularly to ensure that they are functioning correctly and can effectively sterilize items.
Here are five things to consider when validating an autoclave: